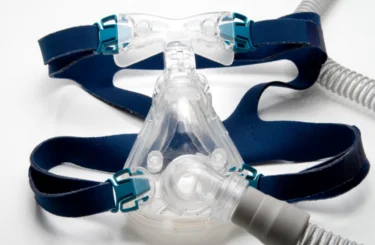
Major CPAP Machine Recall: Philips Devices Endanger Users, May Cause Cancer
Generating summary...
AI can make mistakes and some summarized content may not be accurate.
Nearly 22 million Americans suffer from Obstructive Sleep Apnea (OSA), a sleep disorder involving intermittent airway blockages during sleep. This condition leads to chronic poor sleep and can lead to high blood pressure, chronic heart failure, atrial fibrillation, stroke, and other cardiovascular problems. According to the American Sleep Apnea Association, it is also associated with type II diabetes and depression. The persistent drowsiness of sufferers contributes to many traffic accidents and accidents with heavy machinery.
For many sufferers of OSA, a device called a CPAP (continuous positive airway pressure) machine can provide significant relief. Other patients may benefit from using bilevel positive airway pressure (BiPAP) devices, a similar breathing support machine.
Unfortunately, one of the largest manufacturers of breathing devices recently issued a massive product recall of a number of its most popular machines, including many CPAP and BiPAP models. The FDA issued a statement on June 30, 2021, listing the specific Philips Respironics devices subject to the recall. It identifies numerous models manufactured between 2009 and April 26, 2021, including:
- DreamStation ASV
- DreamStation ST, AVAPS
- SystemOne ASV4
- C-Series ASV
- C-Series S/T and AVAPS
- OmniLab Advanced+
- SystemOne (Q-Series)
- DreamStation
- DreamStation Go
- Dorma 400
- Dorma 500
- REMstar SE Auto
In these defective devices, a polyester-based polyurethane (PE-PUR) sound abatement foam, used to reduce sound and vibration, may physically break down and enter the device’s airstream. This could put users at risk of breathing in or swallowing black debris from the foam or inhaling chemicals released by the breakdown. According to the FDA, the potential risks of exposure to these bits of debris include:
- Irritation to the skin, eye, and respiratory tract.
- Inflammatory response.
- Headache.
- Asthma.
- Toxic or carcinogenic effects to organs, such as kidneys and liver.
The potential risks of exposure to the chemicals that may be released into the device’s air pathway as the PE-PUR foam degrades include:
- Headache.
- Dizziness.
- Irritation in the eyes, nose, respiratory tract, and skin.
- Hypersensitivity.
- Nausea and vomiting.
- Toxic and carcinogenic effects.
Along with the immediate dangers posed by the particulate matter and chemicals, the potential long-term effects of exposure may result in infections, chronic lung or sinus problems, cancer, and other potentially fatal conditions.
The complete list of recalled devices can be found in the Philips Respironics recall notice. Philips is working on manufacturing and distributing repair and replacement kits for the affected machines, but this will take a significant amount of time. Nearly two million recalled devices are currently in use in the United States alone (with nearly two million more in use internationally).
For many users, the damage may already be done. Over 40 lawsuits have been filed on behalf of patients who have suffered injuries from these defective devices. If you use one of the identified machines and have experienced adverse effects like those listed above, you should contact the experienced personal injury and product liability attorneys at Sommers Schwartz. You may be entitled to compensation for your injuries. Contact us today for a free, no-obligation consultation.