FDA Finally Steps Up Measures to Protect Patients from Dangers of All-Metal Hip Implants
Generating summary...
AI can make mistakes and some summarized content may not be accurate.
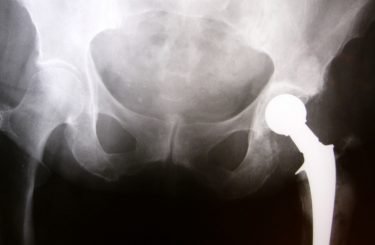
As we’ve reported in earlier posts, an estimated 500,000 patients have undergone surgery to receive a type of artificial hip device known as a “all-metal” or “metal-on-metal” implant. In many cases, these devices have failed when the components – a ball and cup that replicates one’s hip bones – rub against one another and release metallic debris into the surrounding area, causing severe tissue and bone damage.
Many have blamed the U.S. Food & Drug Administration for lax oversight during the approval process for these devices, which only required manufacturers to demonstrate that new designs resembled ones currently on the market and without the need for clinical research. As a result, just a few years ago, all-metal implants were used in one of every three joint replacement procedures in the US, according to the New York Times.
This week, however, the FDA is proposing new guidelines that would require manufacturers of all-metal hip implants to prove the devices are safe and effective before they could continue selling existing models or secure approval for new all-metal designs.
The FDA is expected to provide guidance to physicians and patients, recommending “that patients who are experiencing pain or other symptoms that indicate possible device failure undergo routine testing to detect levels of metallic ions in their blood,” and that “physicians should monitor such tests over time and use that data, along with other information” to make decisions regarding implant replacement.
The injuries that can result from a defective metal-on-metal implant can be serious and disabling. If you have a metal-on-metal implant manufactured by the DePuy division of Johnson & Johnson, Wright Technologies, Smith and Nephew, Stryker Rejuvanate or another company, and are experiencing difficulties with pain or mobility, we encourage you to contact us to discuss your situation.