First Trial in Actos “Pilot Bellwether Program” Set to Begin
Generating summary...
AI can make mistakes and some summarized content may not be accurate.
The first bellwether trial against the maker of the prescription medication Actos will begin later this month in Louisiana, with many awaiting the outcome and its impact on other cases involving the allegedly dangerous drug.
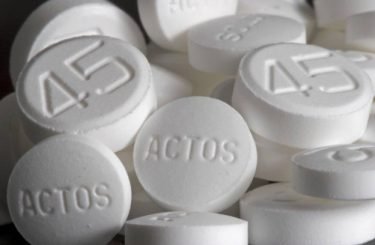
Actos is an anti-diabetic drug that is prescribed to help control blood pressure in people with type 2 diabetes. In 2011, the FDA issued a warning that people who take Actos for extended periods of time may experience an increased risk of developing bladder cancer.
According to lawsuits filed against Takeda Pharmaceutical Co, the manufacturer of Actos (pioglitazone), the drug maker made more than $16 billion on the name brand version of the drug before it went generic in 2012. Attorneys allege that the company knew of the drug’s links to bladder cancer for nearly seven years before alerting the FDA.
Cases against Takeda have been centralized in front of United States District Court Judge Rebecca Doherty in the Western District of Louisiana. The upcoming bellwether trial will be the first of many that will help determine the outcome of thousands of cases that have been filed against the company.
If you have taken Actos and developed bladder cancer, contact Sommers Schwartz today to determine whether you may have a claim.
Jason Thompson is a nationally board certified trial attorney and co-chairs Sommers Schwartz’s Complex Litigation Department. He has a formidable breadth of litigation experience, including class action and multidistrict litigation (MDL), and practices nationwide in both state and federal courts.